- Blog
- About
- Contact
- Mgsv cloaked in silence
- 2020 irs tax tables
- Universe sandbox 2 steam cd key
- Mindview 6-0
- Serial quick pallet maker 4-8-2
- Animosity shows sign letting up
- Jax sheets
- Calendarmenu
- Speech to text in word
- Reactivity series of metals
- Mcafee total protection unlimited devices
- Appzapper 2 serial
- Omnigraffle flowchart tutorial
- Parallels toolbox not activated on new mac
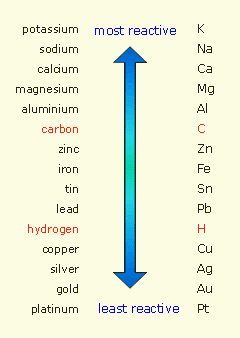
Reactivity series of metals series#
Metals and Alloys Iron Carbon LEGEND: Pure Iron Metal Steel (Alloy) Using the diagram above, explain why alloys are harder and stronger than pure metals? In alloy, the layers of atoms cannot slide over each other easily A mixture of 2 or more metalsĬhapter 9:METALS METALS: The Reactivity SeriesĪt the end of the lesson, students should be able to Describe the reactions, if any between some metals (in the reactivity series) with water Describe the reactions, if any between some metals (in the reactivity series) with steam Describe the reactions, if any between some metals (in the reactivity series) with dilute acid Rank the metals in order of their reactivity Objectives (1)ĭescribe the reduction reactions, if any, of the metal oxides with carbon Describe the reduction reactions, if any, of the metal oxides with hydrogen Describe the action of heat on the metal carbonates and relate thermal stability to the reactivity series At the end of the lesson, students should be able to Objectives (2)
Physical properties of metals METALS Solid state at room temp Shiny appearance High density Good heat conductors Good conductors of electricity High Melting and Boiling point Ductile and malleable Strong and tough Except Mercury (Liquid) Due to strong forces (metallic) except Mercury and alkali metals Due to the presence of mobile ions Can be bent and stretched without breaking Atoms are closely packed Due to the presence of mobile ions

Last Lesson… METALS: The Physical Properties of Metals
